what would be the best way to predict the probability of a baby having cystic fibrosis
Abstract
Purpose: Although neonatal screening (or newborn screening) for cystic fibrosis (CF) is usually practiced, systematic methods for accurate risk calculations are currently lacking.
Methods and Results: We evaluated characteristics of the immunoreactive trypsinogen (IRT) test using the published information. The probability that a neonate has a positive IRT test, if the neonate is affected, a carrier, or a noncarrier, is ≈ 1, 0.041, or 0.011, respectively. We provide methods to calculate genetic risks for a variety of commonly encountered scenarios in which neonates are positive by the IRT test.
Determination: Our Bayesian methods allow CF disease probabilities to be calculated accurately, taking into account all relevant information.
Chief
Cystic fibrosis (CF; OMIM no. 219700) is the almost mutual severe autosomal recessive disorder in Caucasians, affecting ≈ 1 in 2500 live births, and with a carrier frequency of ≈ ane in 25.one CF is caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) cistron [OMIM no. 602421; 219700 (CF); CFTR mutation database, http://www.genet.sickkids.on.ca/cftr/]. More than 1000 unlike CFTR variants have been reported, with allele frequencies varying by ethnic group. Gamble assessment is an essential component of genetic counseling and testing, and Bayesian assay plays a central role in complex risk calculations.ii–5 Nosotros previously developed Bayesian methods to calculate the adventure of an autosomal recessive disease when only one or no mutation is detected in the disease cistron, and another, independent take a chance factor is present (e.g., fetal echogenic bowel as a risk factor for CF).6 Our methods are useful in the setting of CF carrier screening and prenatal testing for CFTR mutations.half dozen We also developed methods to summate CF disease and carrier risks in a diversity of prenatal and carrier screening scenarios that create mutual, difficult issues.vii
Neonatal screening for cystic fibrosis is unremarkably performed as a part of expanded neonatal screening, and ordinarily consists of the immunoreactive trypsinogen (IRT) test followed by CFTR mutation testing if the IRT exam is positive.8 Even so, systematic methods are lacking for accurately calculating CF risks after a positive IRT exam result and various other CFTR test results are obtained. This is because at that place are considerable differences in CFTR allele frequencies among unlike ethnic groups,nine,10 and published information regarding IRT assay characteristics are complex.eleven–xiv In this article, we evaluated published information to calculate the assay characteristics of the IRT test, and provide Bayesian methods for authentic CF risk calculations using concrete scenarios. Our Bayesian methods let CF disease probabilities to be calculated accurately, taking into account all relevant information. These methods can be modified for many unlike scenarios, including scenarios apropos other autosomal recessive disorders. We also discuss the utility of sweat chloride testing after neonatal screening.
METHODS AND RESULTS
There are three central steps to performing a Bayesian risk analysis. First, the problem must exist set out in a format that allows the necessary calculations to be specified. 1 approach is to use probability tables, every bit presented and illustrated in our previous articleshalf dozen,7; an alternative is to graphically depict all pedigree structures and other available information, as in the unified arroyo described by Hodge.xv Second, the appropriate prior probabilities and provisional probabilities must exist determined and included in the table or pedigree. Probabilities relevant for Bayesian analysis in the setting of newborn screening for CF are derived and provided beneath and in Table 1. The third stride involves the mathematical calculation of joint and posterior probabilities.
Frequencies of positive IRT tests amidst CF affected, carrier, and noncarrier neonates
The frequencies of positive IRT tests among CF affected, carrier, and noncarrier newborns were calculated using a single large study.xiv All other published studies on the frequencies of positive IRT tests among newborns did non determine the frequencies of positive IRT tests separately among CF affected, carrier, and noncarrier newborns.11–13,16 Figure 1 illustrates the numbers derived from the data of Scotet et al.14 to calculate each conditional probability that a newborn has hypertrypsinogenemia if the newborn is affected by CF, a carrier, or a noncarrier. In the study by Scotet et al.,14 a cut-off level of IRT was set up equally 600 μg/L, and 60 CF afflicted newborns were detected amongst 160,019 newborns screened, leading to a disease allele frequency of 0.0194 (q 2 = 60/160,019) assuming Hardy-Weinberg equilibrium. Using the iipq (= 0.0380) carrier frequency, approximately six,077 newborns were carriers amongst 160,019 newborns. Amidst one,964 hypertrypsinogenemic newborns, 213 were establish to be carriers with mutations detected by analysis of three exons (exons 7, x, and 11, which contain 85% of CFTR mutations in the Brittany region).14 Thus, approximately 250 (= 213 × 1/0.85) carriers and approximately 1,654 (= one,964 − lx − 250) noncarriers were present among the 1964 hypertrypsinogenemic newborns. The number of CF newborns amongst newborns with IRT levels inside normal limits was not determined.xiv However, the number of CF newborns with normal IRT levels should be small-scale, considering that the CF incidence of threescore/160,019 (= 1/2,667) in this written report was so close to the 1/2,500 incidence widely used as the incidence of CF amongst non-Hispanic Caucasians.1 Therefore, the probability that a CF afflicted, carrier, or noncarrier newborn has hypertrypsinogenemia is estimated to be 1 (= sixty/60), 0.041 (= 250/6,077), or 0.011 [= 1654/(160,019 − lx − half dozen,077)], respectively (Fig. ane). We tin utilize these probabilities every bit conditional probabilities in our Bayesian analyses to calculate CF risks for newborns with a positive IRT examination.
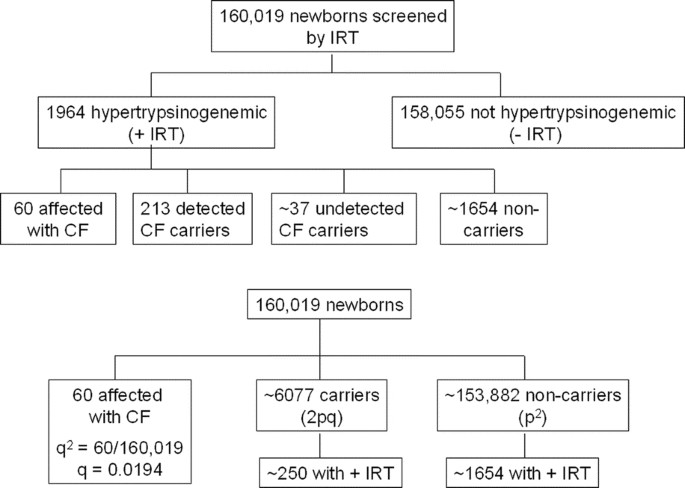
Assay of characteristics of IRT examination.
Bayesian analysis to summate CF risks for neonates with a positive IRT test
A fraction of each major CFTR disease allele amid all CFTR disease alleles and a mutation detection charge per unit are summarized for each of v major ethnic groups (Table i). Nosotros assumed that CFTR mutation testing detects the 23 CFTR mutations designated for cancer screening past the American College of Medical Genetics (ACMG),x which is a slight modification of the original 25 CFTR mutation panel.1,17 We used carrier frequencies for not-Hispanic Caucasian (ane/25), Ashkenazi Jewish (1/25), African American (i/65), Hispanic Caucasian (1/46), and Asian American (ane/90).i,6 We used mutation detection rates and the fractions of major mutations for major ethnic groups as reviewed previously.10 In particular, we used mutation detection rates of 0.88, 0.94, 0.64, and 0.72 for non-Hispanic Caucasian, Ashkenazi Jewish, African American, and Hispanic Caucasian populations, respectively (Table 1). Ane can modify our methods with regard to mutation detection rates, as long equally the fraction of each mutation in a CFTR testing panel in question among all CFTR disease alleles is known. The ethnicity-specific fractions of the common F508del mutation [C.1522_1524 del TTT (p.Phe508del)] amidst all CFTR mutations are 0.72, 0.31, 0.44, and 0.54 for non-Hispanic Caucasian, Ashkenazi Jewish, African American, and Hispanic Caucasian populations, respectively (Table 1).
Scenario 1. Non-Hispanic Caucasian neonate with a positive IRT test, CFTR mutation testing on the neonate awaiting or not performed, and no family history or CFTR mutation testing on parents or other relatives
There is commonly a time delay between the time of a positive IRT test result and the time of the completion of CFTR mutation testing. What is the gamble that the neonate is affected before CFTR mutation testing, or if CFTR mutation testing is not performed? Suppose a non-Hispanic Caucasian neonate has a positive consequence in the IRT examination; the Bayesian analysis for the CF risk of the neonate is shown in Table 2. Earlier the IRT exam is performed, the probability that the neonate is afflicted, a CF carrier, or a noncarrier is 0.0004, 0.04, or 0.96, respectively. The conditional probability that the neonate is positive by the IRT test, if the neonate is affected, a carrier, or a noncarrier, is 1, 0.041, or 0.011, respectively. The joint probability that the neonate is affected, a carrier, or a noncarrier, and that the neonate shows a positive IRT effect, is the production of each prior probability and the conditional probability for each column (i.e., "Affected," "Carrier," or "Noncarrier"). To obtain the posterior probability that the neonate is affected given a positive IRT test, one can divide the articulation probability that the neonate is affected and shows a positive IRT exam, past the probability that the neonate shows a positive IRT examination regardless of genotype (i.e., the sum of all of the joint probabilities), yielding 0.032. The posterior probability that the neonate is a carrier or a noncarrier given a positive IRT test is the joint probability for each column divided by the sum of all of the joint probabilities (0.13 or 0.84, respectively). After CFTR mutation testing is performed, Table 2 will be modified to comprise the results of CFTR mutation testing (see Scenarios two and 3).
Scenario 2. Non-Hispanic Caucasian neonate with a positive IRT test, one identifiable CFTR mutation (F508del), and no family history of CF
As in Scenario one, the probability (before IRT and CFTR mutation testing) that the neonate is affected, a CF carrier, or a noncarrier is 0.0004, 0.04, or 0.96, respectively. The Bayesian analysis for this scenario is shown in Table 3. The challenge in this instance is in the 2nd step of the process, which is determining the conditional probability of finding a single identifiable mutation, for each column. We demonstrate the process for the outset column: if the neonate is affected, then both parents are known to be carriers, and at that place are two possibilities, which are that the mutation from parent A (Da) is F508del with the mutation from parent B (Db) undetectable, or the mutation from parent B (Db) is F508del with the mutation from parent A (Da) undetectable. The probability in this indigenous group that a CFTR mutation is F508del or undetectable can exist determined from Table 1. Thus, the conditional probability that the neonate has only one identifiable mutation F508del if afflicted is the fraction of the identified mutation (amongst all CFTR mutations) in the male parent's ethnic background population multiplied past (1 − mutation detection charge per unit for the mother's ethnic background population), plus the fraction of the identified mutation (amongst all CFTR mutations) in the female parent's ethnic background population multiplied by (1 − mutation detection charge per unit for the father's ethnic background population) (i.e., 0.72 × 0.12 + 0.12 × 0.72 = 0.1728). For the 2d column in Table three, the conditional probability that the neonate has one identifiable mutation if the neonate is a carrier is the fraction of the identified mutation in the parents' groundwork population (i.e., 0.72). Clearly, for the tertiary cavalcade in Table 3, the conditional probability that the neonate has one identifiable mutation F508del if the neonate is a noncarrier is 0. By applying the 3rd footstep of carrying out the necessary mathematical calculations, it is determined that the posterior probability that the neonate is affected is 0.056.
Scenario 3. Non-Hispanic Caucasian neonate with a positive IRT test, no identifiable CFTR mutation, and no family history of CF
This scenario is probably one of the most mutual counseling scenarios in CF newborn screening. As in Scenarios one and 2, the prior probability that the neonate is afflicted, a CF carrier, or a noncarrier is 0.0004, 0.04, or 0.96, respectively. The Bayesian assay for this scenario is shown in Table 4. The conditional probability that the neonate has no identifiable mutation, if the neonate is affected, a CF carrier, or a noncarrier, is (1 − mutation detection rate for the father's ethnic background population) multiplied by (1 − mutation detection charge per unit for the female parent'south ethnic background population), (1 − mutation detection rate for the parent's indigenous groundwork population), or 1, respectively. The posterior probability that the neonate is affected or a carrier is 0.00054 or 0.018, respectively.
Scenario iv. Neonate with a non-Hispanic Caucasian parent A, an Ashkenazi Jewish parent B, a positive IRT test, 1 identifiable CFTR mutation (F508del), and no family unit history of CF
For this scenario, we cannot begin with the prior probabilities of particular hypotheses for the fetus as in Scenarios ii and 3. Instead, we must begin with the prior probabilities of particular hypotheses for the parents earlier the IRT test and the CFTR mutation testing are performed. This is considering the ethnicities of parent A and parent B are dissimilar, as are the CFTR allelic spectra in these ethnicities, so we need to follow paternally-derived and maternally-derived mutations separately if whatsoever mutation exists in either parent. The Bayesian analysis for this scenario is shown in Tabular array 5, which is based on Scenario 1 in Ogino et al.6 For simplicity, columns for which the neonate is a noncarrier have non been included in this table; this is considering the joint probability for each of these columns volition be 0, considering the conditional probability that a noncarrier neonate has a single identifiable mutation is 0. Before the IRT examination and CFTR mutation testing are performed, the probability that parent A or parent B is a CF carrier is 0.04 or 0.04, respectively. For each column, the conditional probability given the parental genotypes that the neonate is affected, a carrier with a mutation from parent A, or a carrier with a mutation from parent B tin can be calculated by applying simple Mendelian ratios. For example, if both parents are carriers, these three conditional probabilities are one/4, 1/4, or 1/4, respectively. The conditional probabilities of a positive IRT test result given each column are incorporated in the next row of the table, as before. Adjacent, we incorporate the information that the neonate has one identifiable mutation F508del. If the neonate is affected, there are two possibilities: either the mutation from parent A (Da) is F508del and the mutation from parent B (Db) is undetectable, or the mutation from parent B (Db) is F508del and the mutation from parent A (Da) is undetectable. Columns A and B in Tabular array v correspond to these two possibilities. The posterior probability that the neonate is affected is the sum of the posterior probabilities of columns A and B, or 0.037.
Scenario four provides an example of how the unified arroyo described by Hodgefifteen can exist used to perform Bayesian take chances calculations (Fig. ii). In this arroyo, nosotros place all possible pedigree configurations that could lead to a neonate with a positive IRT test and ane F508del (ΔF508) mutation. The six possible configurations are depicted in a schematic style rather than in a table. The relevant probabilities are drawn directly onto the pedigree. The articulation probability of each configuration is determined, allowing for the calculation of posterior risk that the neonate is affected. The main reward of this method is that it allows the identification of all possible pedigree permutations with relative ease, and that it is clear that the Bayesian analysis begins at the pinnacle of the pedigree.
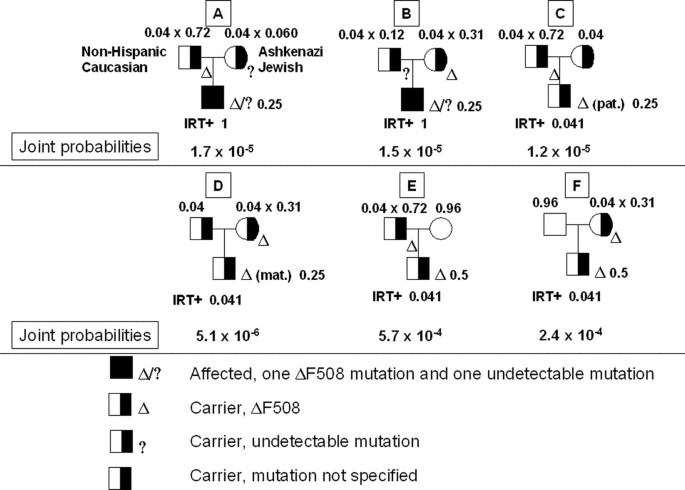
Unified approach to Scenario iv. Starting at the superlative of the pedigree, the probabilities and data for each individual are noted. The joint probability for configuration C, for example, i.due east., the probability that the neonate is a carrier with a paternally-derived ΔF508 and a positive IRT test, is the product of the probabilities that the begetter is a carrier (0.04) and has ΔF508 given his carrier status (0.72); that the mother is a carrier (0.04); that the neonate has inherited ΔF508 from the father and a normal allele from the mother (1/4); and that the neonate tests positive for IRT given his or her carrier condition (0.041).
Scenario v. Neonate of a Non-Hispanic Caucasian parent A and an African American parent B, with a positive IRT test, no identifiable CFTR mutation, and no family history of CF
Before the IRT test and CFTR mutation testing are performed, the probability that parent A or parent B is a CF carrier is 1/25 or 1/65, respectively. The Bayesian analysis for this scenario is shown in Table 6, which is based on Scenario 5 in Ogino et al.vi The provisional probabilities that the neonate has no identifiable mutation given that the neonate is afflicted, a carrier of a mutation inherited from parent A, a carrier of a mutation inherited from parent B, or a noncarrier are calculated from Table ane (every bit in Scenario 3). The conditional probability that the neonate has no identifiable mutation if affected is (1 − mutation detection charge per unit for parent A'southward indigenous groundwork population) multiplied by (1 − mutation detection charge per unit for parent B'southward indigenous groundwork population). The conditional probability that the neonate has no identifiable mutation if the neonate is a carrier with a mutation derived from parent A or B is (1 − mutation detection charge per unit for parent A's ethnic background population) or (1 − mutation detection rate for parent B's ethnic background population), respectively. The posterior probability that the neonate is afflicted is 0.00061 (= ane/1600). The posterior carrier probability of the neonate is the sum of the posterior probabilities of columns B, C, East, and Thou, or 0.019.
Scenario 6. Neonate of a Non-Hispanic Caucasian parent A, a Hispanic American parent B, with a positive IRT test and one identifiable CFTR mutation (F508del); parent A with a family history of CF, parent B with no family unit history of CF, and neither parent tested
Because parent A has a family history of CF, prenatal testing and/or carrier screening should have been offered. Unfortunately, however, this scenario is not uncommon. Suppose that parent A has an affected nephew who has not been tested, and that neither of the parents has been tested. Before the IRT test and CFTR mutation testing are performed, the probability that parent A or B is a CF carrier is 1/2 or 1/46, respectively. The Bayesian analysis and the unified arroyo (Hodge15) are shown in Table 7 and Figure 3. This example is based on Scenario 1 in Ogino et al.6 At that place is a small possibility that each parent of parent A has a disease allele, and that parent A inherited the disease allele different from the one present in the affected nephew. Including this possibility, parent A's carrier probability is 0.5025, representing merely a 0.5% increase from the 0.five carrier probability that derives from the mutations present in the afflicted nephew (calculations non shown). Our approach starting from parent A is straightforward and the results are substantially the same every bit those by an approach starting from parents of parent A (calculations not shown). The prior probability that parent A is a carrier or a noncarrier is i/2 or 1/2, respectively. The prior probability that parent B is a carrier or a noncarrier is 1/46 or 45/46, respectively. The conditional probability that the neonate is affected, a carrier with a mutation from parent A, or a carrier with a mutation from parent B, if both parents are carriers, is ane/iv, 1/4, or 1/4, respectively. The conditional probability that the neonate is a carrier with a mutation from parent A (or parent B), if parent A is a carrier (or a noncarrier) and parent B is a noncarrier (or a carrier), is 1/2 (or one/two, respectively). If the neonate is afflicted, at that place are two possibilities: either the mutation from parent A (Da) is F508del and the mutation from parent B (Db) is undetectable, or the mutation from parent B (Db) is F508del and mutation from parent A (Da) is undetectable. Thus, the conditional probability that the neonate has 1 identifiable mutation if affected is the sum of the fraction of that identified mutation in parent A's ethnic background population multiplied by (i − mutation detection rate for parent B's ethnic background population) and the fraction of that identified mutation in parent B'due south indigenous background population multiplied by (1 − mutation detection rate for parent A's ethnic background population) (i.e., 0.72 × 0.28 + 0.54 × 0.12). The conditional probability that the neonate has one identifiable mutation if the neonate is a carrier due to a mutation derived from parent A or parent B is the fraction of that identified mutation in that parent's groundwork population (i.e., 0.72 or 0.54, respectively). The posterior probability that the neonate is affected is the sum of the posterior probabilities of columns A and B, or 0.088.
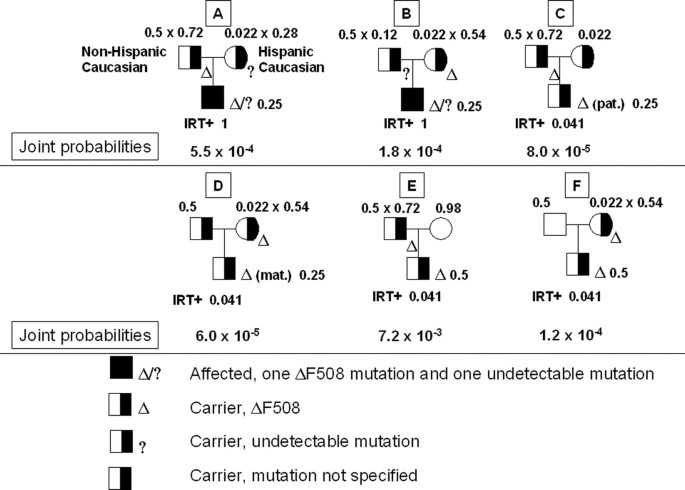
Unified approach to Scenario half dozen. Starting at the tiptop of the pedigree, the probabilities and information for each private are noted. Joint probability for configuration A, for example, i.e., the probability that the offspring is affected with a paternally-derived ΔF508 and a maternally-derived, undetectable mutation, and a positive IRT test, is the product of the probabilities that the male parent is a carrier (one/2) and has ΔF508 given his carrier status (0.72; the fraction of ΔF508 amidst all of non-Hispanic Caucasian mutations); that the mother is a carrier (0.022; the carrier frequency in the Hispanic population) and has an undetectable mutation given her carrier status (0.28; i.e., 1 − mutation detection charge per unit in the Hispanic population); that the neonate has inherited ii mutations (1/4) given such carrier parents; and that the neonate tests positive for IRT given his or her affected status (i).
Scenario seven. Neonate of a Non-Hispanic Caucasian parent A and a Hispanic American parent B, with a positive IRT examination and no identifiable CFTR mutation; parent A with a family history of CF and negative CFTR carrier screening; parent B with no family history of CF and no carrier screening performed
The Bayesian analysis for this scenario is shown in Tabular array 8, which is based on Scenario vi in Ogino et al.6 Suppose that parent A has an affected nephew who has not been tested. Before the IRT examination and whatever CFTR mutation testing are performed, the probability that parent A or parent B is a CF carrier is 1/2 or 1/46, respectively. The conditional probability that parent A tests negative is (ane − mutation detection rate for parent A's indigenous background population). The provisional probability that the neonate has no identifiable mutation if afflicted is (1 − mutation detection charge per unit for parent B'due south ethnic background population) × i, because parent A tests negative and a mutation derived from parent A, if present, is undetectable. The conditional probability that the neonate has no identifiable mutation if the neonate is a carrier with a mutation derived from parent A or parent B is 1 or (one − mutation detection charge per unit for parent B's ethnic background population), respectively. The posterior probability that the neonate is affected is 0.013. The posterior carrier probability for the neonate is the sum of the posterior probabilities of columns B, C, Eastward, and Thou, or 0.xviii.
Scenario 8. Non-Hispanic Caucasian neonate with a positive IRT test and i identifiable CFTR mutation (F508del) present in parent A; parent B with no family unit history of CF and negative CFTR carrier screening
Earlier the neonate was tested, the probability that parent B is a carrier is 0.04. The Bayesian assay for this scenario is shown in Table 9, which is based on Scenario iv in Ogino et al.half-dozen The conditional probability that parent B tests negative is (1 − mutation detection rate for parent B'south ethnic background population). The conditional probability that the neonate has one identifiable mutation if affected is 1, because a mutation derived from parent A, if present, is identifiable, and a mutation derived from parent B, if present, is undetectable. The provisional probability that the neonate has ane identifiable mutation, if the neonate is a carrier with a mutation derived from parent A or parent B, is 1 or 0, respectively, because Db, if present, is undetectable. The posterior probability that the neonate is affected is 0.057.
Scenario ix. Neonate with a positive IRT test, one or no identifiable CFTR mutation (F508del), and a positive or negative sweat test
After neonatal screening with a positive IRT exam and no detectable CFTR mutation, whether a sweat examination is performed depends on the neonate'south adventure and other factors. If the IRT test is positive and i detectable mutation is present, the neonate typically undergoes confirmatory sweat testing. Sweat chloride levels in infants with CF, CF carrier infants, and noncarrier infants in Wisconsin were adamant by Farrell and Koscik.18 The mean ± 3 SD of sweat chloride in noncarrier infants (Due north = 184) and in carrier infants (N = 128) ranged from −5.0 to 26.2 mEq/L and from −10.three to forty.1 mEq/50, respectively.18 Mean ± 3 SD of sweat chloride in CF infants with homozygous F508del (Due north = 61), in CF infants with one F508del and one non-F508del mutation (Due north = 47), and in CF infants with two non-F508del mutations (N = 7) ranged from 72.4 to 127.half dozen mEq/L, from 52.0 to 143.2 mEq/L, and from 57.3 to 131.9 mEq/Fifty, respectively.18 Therefore, when properly performed, sweat testing can discriminate CF infants from not-CF infants, with nigh no overlap of sweat chloride levels between these two groups. Notwithstanding, LeGrys's report19 of proficiency testing surveys on sweat analysis for CF demonstrated that some laboratories reported erroneous results. One should interpret sweat-test results cautiously, particularly when clinical findings are inconsistent with them, or when the sweat test is performed on neonates other than non-Hispanic Caucasians. To confirm that the sweat test can be a confirmatory test among ethnicities other than non-Hispanic Caucasians, ethnicity-specific sweat chloride levels amid CF neonates, CF carrier neonates, and noncarrier neonates need to be adamant. Sweat chloride levels does non seem to predict the severity of lung disease amid CF patients.xx
Discussion
Bayesian analysis plays an essential role in calculations of genetic risk.2–v,21 We previously developed Bayesian methods to summate autosomal recessive disease risks with only one or no detectable mutation,6 and methods to summate CF disease and carrier risks in a variety of prenatal and carrier screening scenarios that provide common, hard problems.7 These methods are specially useful for CF. Using concrete examples, we have illustrated herein principles of genetic chance calculations for a multifariousness of common scenarios that ascend in CF neonatal screening.
Although prenatal screening and carrier screening for all couples take been advocated by the American Higher of Medical Genetics (ACMG) and the American Higher of Obstetrics and Gynecology (ACOG),1,17,22 a large number of pregnancies and couples are not screened earlier birth, because of economic or other reasons. Neonatal screening is relatively easy to perform, and has been shown to be an effective way to prevent malnutrition and improve long-term growth of neonates affected with CF.23
The assay characteristics of the IRT examination that we used here were derived from a single large study on newborn screening by the IRT test,14 because this study is the just report bachelor that allows calculations of the probability of a positive IRT test if a neonate is affected, a carrier, or a noncarrier. However, normal IRT value ranges may exist unlike in different ethnic groups. Ethnicity-specific assay characteristics of the IRT test should be used when comprehensive information on IRT exam results for CF affected, carrier, and noncarrier neonates in unlike indigenous groups are bachelor in the future.
Effects of prenatal ultrasound screening, prenatal CFTR mutation testing, and neonatal screening on the birth prevalence of CF in Brittany, France have been investigated and an approximately 30% decrease in CF prevalence was estimated.24 Effects of public health measures on the frequencies of the various CFTR disease alleles need to exist investigated, because they will impact risk assessments for CF considerably.
In conclusion, nosotros present a variety of Bayesian methods to calculate CF risks for neonates with a positive IRT test. Our methods are applicative to many common clinical scenarios and allow CF disease risks to exist calculated accurately, taking into account all relevant information. Our methods tin also be applied to other autosomal recessive disorders and screening scenarios.
NOTES ADDED IN PROOF
- i
In exercise, when complicated scenarios are encountered, the calculations should always exist checked independently by another geneticist to guard against errors; The authors abide by this dominion.5
- 2
On March 8, 2005, The ACMG report entitled "Newborn Screening: Toward a Uniform Screening Panel and System" was submitted to the Health Resources and Services Administration of the U.South. Section of Health and Man Services, and fabricated available for public comment. Among the 29 weather for which there was a sufficient scientific and medical basis to recommend inclusion in newborn screening panels was cystic fibrosis. Its inclusion is similarly advocated past the March of Dimes and the Cystic Fibrosis Foundation.
References
-
Richards CS, Bradley LA, Amos J, Allitto B, Grody WW, Maddalena A et al. Standards and guidelines for CFTR mutation testing. Genet Med 2002; 4: 379–391.
-
Young ID . Introduction to take chances adding in genetic counseling, 2nd ed. Oxford: Oxford Academy Press, 1999.
-
Bridge PJ . The adding of genetic risks: Worked examples in DNA diagnostics, 2nd ed. Baltimore: The Johns Hopkins Academy Press, 1997.
-
Ogino S, Wilson RB . Bayesian analysis and risk cess in genetic counseling and testing. J Mol Diagn 2004; 6: one–9.
-
Hodge SE, Flodman PL . Take a chance calculations: Even so essential in the molecular age. Am J Med Genet 2004; 129A: 215–217.
-
Ogino S, Wilson RB, Grody WW . Bayesian chance cess for autosomal recessive diseases: Fetal echogenic bowel with one or no detectable CFTR mutation. J Med Genet 2004; 41: e70.
-
Ogino Due south, Wilson RB, Gold B, Hawley P, Grody WW . Bayesian analysis for cystic fibrosis risks in prenatal and carrier screening. Genet Med 2004; half dozen: 439–449.
-
Southern KW, Littlewood JM . Newborn screening programmes for cystic fibrosis. Paediatr Respir Rev 2003; four: 299–305.
-
Bobadilla JL, Macek M, Jr, Fine JP, Farrell PM . Cystic fibrosis: A worldwide assay of CFTR mutations–correlation with incidence information and awarding to screening. Hum Mutat 2002; 19: 575–606.
-
Watson MS, Cutting GR, Desnick RJ, Driscoll DA, Klinger K, Mennuti M et al. Cystic fibrosis population carrier screening: 2004 revision of american college of medical genetics mutation panel. Genet Med 2004; 6: 387–391.
-
Massie J, Du Sart D, Forshaw Grand, Carlin J, Forrest SM . The relationship betwixt neonatal immunoreactive trypsinogen, deltaf508, and ivs8-5t. J Med Genet 2000; 37: 629–632.
-
Gregg RG, Simantel A, Farrell PM, Koscik R, Kosorok MR, Laxova A et al. Newborn screening for cystic fibrosis in Wisconsin: Comparison of biochemical and molecular methods. Pediatrics 1997; 99: 819–824.
-
Corbetta C, Seia G, Bassotti A, Ambrosioni A, Giunta A, Padoan R . Screening for cystic fibrosis in newborn infants: Results of a pilot programme based on a 2 tier protocol (irt/Dna/irt) in the Italian population. J Med Screen 2002; 9: 60–63.
-
Scotet 5, De Braekeleer Thousand, Audrezet MP, Lode L, Verlingue C, Quere I et al. Prevalence of CFTR mutations in hypertrypsinaemia detected through neonatal screening for cystic fibrosis. Clin Genet 2001; 59: 42–47.
-
Hodge SE . A simple, unified arroyo to Bayesian hazard calculations. J Genet Couns 1998; 7: 235–261.
-
Scotet V, de Braekeleer M, Roussey Yard, Rault G, Parent P, Dagorne M et al. Neonatal screening for cystic fibrosis in Brittany, France: Cess of 10 years' experience and impact on prenatal diagnosis. Lancet 2000; 356: 789–794.
-
Grody WW, Cutting GR, Klinger KW, Richards CS, Watson MS, Desnick RJ . Laboratory standards and guidelines for population-based cystic fibrosis carrier screening. Genet Med 2001; 3: 149–154.
-
Farrell PM, Koscik RE . Sweat chloride concentrations in infants homozygous or heterozygous for f508 cystic fibrosis. Pediatrics 1996; 97: 524–528.
-
LeGrys VA . Sweat analysis proficiency testing for cystic fibrosis. Pediatr Pulmonol 2000; 30: 476–480.
-
Davis PB, Schluchter MD, Konstan MW . Relation of sweat chloride concentration to severity of lung disease in cystic fibrosis. Pediatr Pulmonol 2004; 38: 204–209.
-
Ogino S, Wilson RB . Genetic testing and risk assessment for spinal muscular atrophy (SMA). Hum Genet 2002; 111: 477–500.
-
Richards CS, Grody WW . Prenatal screening for cystic fibrosis: Past, present and hereafter. Expert Rev Mol Diagn 2004; 4: 49–62.
-
Farrell PM, Kosorok MR, Rock MJ, Laxova A, Zeng Fifty, Lai HC et al. Early on diagnosis of cystic fibrosis through neonatal screening prevents severe malnutrition and improves long-term growth: Wisconsin cystic fibrosis neonatal screening study group. Pediatrics 2001; 107: 1–xiii.
-
Scotet V, Audrezet MP, Roussey Yard, Rault M, Blayau M, De Braekeleer Grand et al. Impact of public health strategies on the birth prevalence of cystic fibrosis in Brittany, France. Hum Genet 2003; 113: 280–285.
Acknowledgements
This projection has been funded in whole or in part with Federal Funds from the National Cancer Institute, National Institutes of Health. We give thanks James Yankaskas and Michael Watson for helpful discussions.
Author information
Affiliations
Corresponding author
Rights and permissions
About this article
Cite this commodity
Ogino, S., Flodman, P., Wilson, R. et al. Risk calculations for cystic fibrosis in neonatal screening by immunoreactive trypsinogen and CFTR mutation tests. Genet Med 7, 317–327 (2005). https://doi.org/10.1097/01.GIM.0000162871.68167.8A
-
Received:
-
Accustomed:
-
Effect Date:
-
DOI : https://doi.org/10.1097/01.GIM.0000162871.68167.8A
Keywords
- Bayesian
- CFTR
- cystic fibrosis
- genetic counseling
- screening
Further reading
Source: https://www.nature.com/articles/gim200565
0 Response to "what would be the best way to predict the probability of a baby having cystic fibrosis"
Postar um comentário